
Contact
- cking@imb.sinica.edu.tw
- (O) 886-2-2782-7854
(L) 886-2-2789-9974 - N211, Institute of Molecular Biology, Academia Sinica
Research
Biology of Prions and Amyloids
How do “protein-only” prions achieve strain variations? And what is the use of a prion? These are some of the questions we would like to answer.
We have demonstrated that strains of the yeast prion [PSI] are differently folded amyloid fibers of the same Sup35 protein. Amyloid is associated with many human neurodegenerative diseases, including the Alzheimer’s disease and the Huntington’s disease. In these conditions, distinct proteins are observed to aggregate and adopt a common cross-β conformation, with β-strands lying perpendicular to the fiber axis. In the case of [PSI], more than 30 different strains of the wild-type Sup35 protein were isolated in our laboratory. Structural characterizations are carried out to understand how the different strains can faithfully self-maintain in spite of sharing the cross-β architecture. While prions are amyloid, the converse is not always true. Complex interplays between amyloid conformations and appropriate cellular co-factors determine whether an amyloid structure can propagate as a prion inside cells. We are creating artificial cellular environments in yeast to elucidate these intricate interactions. The knowledge gained may help explain why β-amyloid and tau filaments spread along predictable cell paths in the brain of Alzheimer’s patients.
Some filamentous fungi use prions to discriminate self from non-self. We are very interested in discovering new prions and elucidating their roles in cellular control. We will also exploit prions to engineer novel activities in a cell. This line of research could lead to novel biotechnological applications.
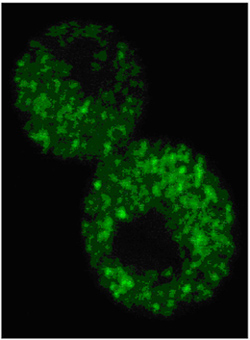
Prion particles roam the cells.
Copyright © 2017 IMBCC. All rights reserved. |