Liuh-Yow Chen Lab

Contact
- lyowchen@gate.sinica.edu.tw
- (O) 886-2-2788-3457
(L) 886-2-2789-9217 - 421, Institute of Molecular Biology, Academia Sinica
Research
Telomere Biology and Telomere Diseases
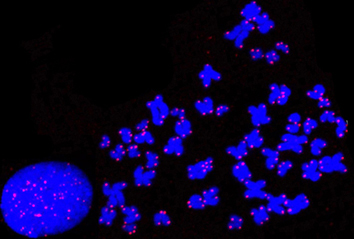
Telomeres are specialized chromatin structures at the distal termini of linear chromosomes essential for genome stability by preventing chromosomes from degradation and fusion. Deregulation of telomere structural maintenance is involved in cancer development and telomere dysfunction causes a number of premature aging diseases, termed telomere syndrome. Our research focus is on understanding the molecular basis of telomere maintenance and its involvement in human diseases.
I. Telomerase regulation
The conventional DNA replication machinery is unable to fully replicate the genome when it reaches the ends of chromosomes. To prevent telomere shortening, telomerase, a ribonucleoprotein enzyme, extends telomeres by reverse transcription. This is crucial for telomere length maintenance in germ cells, stem cells and cancers. Using cancer cells as a model, we found that spatial regulation and cell cycle- dependent phosphorylation of TIN2-TPP1 proteins, which are the components of telomere-associated complex Shelterin, control telomerase recruitment for telomere lengthening. Moreover, we also identified the trimeric protein complex CST (CTC1-STN1-TEN1) as a terminator of telomerase. We found that CST associates with the newly telomerase-extended telomeres and interacts with TPP1-POT1 to limit telomerase action to maintain telomere length homeostasis.
II. CST and telomere syndrome
Mutations in CTC1 cause telomere syndromes, including Coats plus and Dyskeratosis Congenita. Through studying the disease-causing mutations, we identified that mutations can affect the molecular interactions of CTC1 with STN1, DNA Polymerase-alpha, and/or ssDNA. In addition, in vivo, specific CTC1 mutants are defective in nuclear and/or telomeric localizations and their expressions commonly cause telomere replication defects. This study reveals an association of deficient telomere replication with human genetic diseases and could benefit the further development of therapeutic interventions.
III. Telomeres and mitochondria connection
Evidences suggest a functional interaction between telomeres and mitochondria but the mechanism remains elusive. We found that telomeric protein TIN2 has an alternative cellular localization and function in mitochondria. Mitochondrial TIN2 regulates oxidative phosphorylation and energy metabolism in cancer cells. TPP1 interacts with the TIN2 and controls its telomeric and mitochondrial localizations. This study provides a molecular link between telomeres and mitochondria and has implications on the aging process.
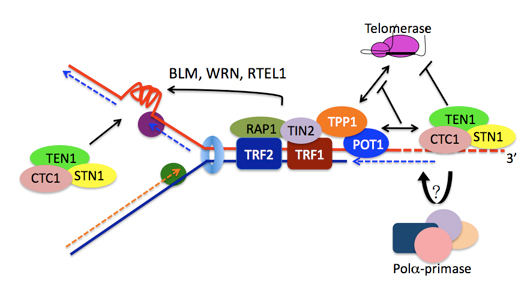
Multiple roles of mammalian CST in telomere replication.
Copyright © 2017 IMBCC. All rights reserved. |