Research
Tolerance and Inflammation in Immunity
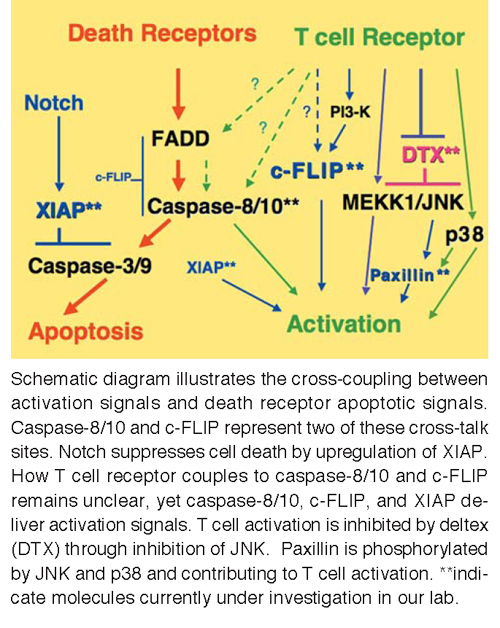
T cell tolerance is maintained by T cell anergy and regulatory T cell (Treg) suppression. We identified a regulatory role of deltex1 in T cell tolerance. Overexpression of deltex1 suppresses T cell activation. We also showed that Dtx1 is a transcription target of NFAT and is up-regulated in T cell anergy. Deficiency of deltex1 augments T cell activation, confers resistance to anergy induction, enhances autoantibody generation, and increases inflammation. Deltex1 also binds PKCθ and promotes PKCθ degradation and, as a consequence, DTX1 increases Cbl-b protein stability. Deltex1 therefore represents a key component in T cell anergy. We further identified a specific role for deltex1 in the maintenance of Foxp3 protein stability and Treg inhibitory activity; deltex1 interacts with HIF-1α and induces HIF-1α degradation. We also identified another tolerance-associated molecule, death associated protein kinase (DAPK). DAPK suppresses T cell activation through targeting to NF-κB. Deficiency in DAPK leads to preferential differentiation of Th17 cells and development of experimental autoimmune encephalomyelitis. We found that Th17 differentiation is accompanied by HIF-1α-mediated DAPK degradation. We are currently delineating the interactive network between deltex1, DAPK, Foxp3, and HIF-1α, and determining how the stability of these proteins controls T cell tolerance and over-activation. We are similarly examining the activation of NF-κB by XIAP through specific protein ubiquitination.
We identified several unexpected molecules that are required for the full activation of inflammasome. Deficiency in PML or c-FLIP selectively reduced the processing of procaspase-1. We further showed that PML is required for the assembly of the NLRP3 inflammasome, but not AIM2 inflammasome. In contrast, c-FLIP is essential for the activation of both NLRP3 inflammasome and AIM2 inflammasome. Our results illustrate that PML and c-FLIP are valid targets for treatment of inflammatory diseases caused by over-activation of inflammasomes.
Copyright © 2017 IMBCC. All rights reserved. |
