Yu-Chan Chao Lab
Research
Transcriptional control and baculovirus surface display for biotechnological applications
Baculoviruses are a group of insect specific pathogens. In our laboratory, we are mainly focus on the study of virus/host interactions in insects, in addition, we have also applied the baculovirus as a tool for manipulating host cells or organism for better basic and application studies. There are two major research studies carried on in our laboratory:
I. Establishment of fluorescent insect by baculovirus for the study of virology, insect physiology, and virus/insect interactions:
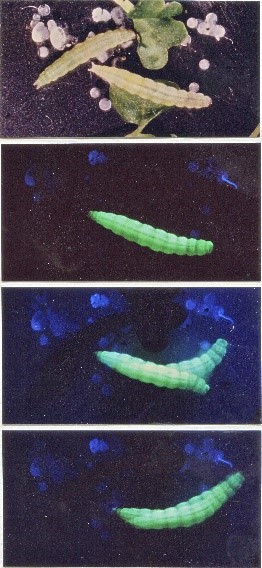
We have successfully generated fluorescent insects by baculovirus vector to trace the movement of this major insect micro-pathogen in insects. We have also engineered fluorescent silkworms to identify the best silkworm strain for protein expression. In order to study virus/mosquito interactions, we have successfully engineered essentially all different mosquito genera using baculovirus as a vector for gene delivery with success. This will be an excellent platform for better mosquito studies. Although these are insect and insect-related viruses, they will significantly improve our understanding of insect physiology, the detection of viral infections, and human pathogenic viruses/mosquitoes interactions in the future for the improvement of global human health.


Fig. 1. Baculovirus-mediated fluorescent insects for the study of virus gene expression and virus/host interactions. Left, the first live fluorescent insect labelling using diamond back moth larvae, top panel: bright light image; bottom three panels: fluorescent larvae for life tracing of virus infection (Chao et al., 1996, Nature 380, 396-397). Middle, fluorescent silkworm larvae, in 2010 Taipei international flora exhibition. Right: fluorescent mosquito for the study of virus/mosquito interactions (Naik et al., 2018, Scientific Reports, 8:17778).
II. Surface display of exogenous membrane proteins on baculovirus and/or insect cells for functional, immunological, and pathological studies:
In year 2003-2004, SARS was a serious international crisis affecting human health, and compounded economic crisis due to its high mortality rate. SARS kills human by cytokine storm, and we provided the first prove that cytokine storm is triggered by the spike protein on the surface of SARS by engineering of spike from SARS onto the membrane of baculovirus (Chang et al., 2004). The critical point for success is that all other laboratories studied purified spike and rendered it non-functional due to the destroy of its membrane anchored trimeric structure, but in our system, the spike was “isolated” on baculovirus envelope, as functional trimeric protein (Fig. 2). This technology has further improved in recent years, and we have engineered the major surface proteins of influenza viruses on baculovirus. Since our first success in anchoring of antigens from detrimental viruses in year 2004 (Chang et al., 2004), we have proved that baculovirus displaying major surface proteins from other viruses can serve as excellent vaccines effective to prevent the infection of viral diseases (Fig. 3), our novel surface protein displayed on baculovirus will be an important asset for the improvement of international human health.
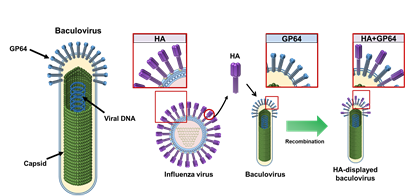
Fig. 2. Foreign viral proteins can be displayed efficiently on the envelope of baculovirus. (A) Schematic representation of the rod-shaped AcMNPV particle. GP64 is the major glycoprotein on the envelope responsible for cell entry and budding of the virus. Capsids are proteins surrounding the viral DNA genome. (B) Display of hemagglutinin (HA) from influenza virus on the baculovirus surface. The derived recombinant virus displays the trimeric HA on the viral surface as well as the native GP64.
Introduction
Baculoviruses are a group of rod-shaped insect-specific viruses, which contains a dsDNA genome of around 80-180 kb. It is a non-human infectious virus and thus safe and easy for manipulation. This virus has long been used for the expression of engineered proteins in insect cells for medical studies and biotechnological applications. It can also be used as a safe and efficient tool for gene delivery in mammalian cells and organisms. In our laboratory, we are not only manipulating baculovirus for the expression of engineered proteins, but also applying it as a versatile tool for molecular biological studies. We have found that baculovirus takes a unique approach using immediate early gene IE2 to target its own DNA indirectly through Daxx, this is the major component of the host defense mechanism. Then, IE2 forms unique hollow sphere-shaped nuclear bodies, which we name clathrate cage-like apparatus (CCLA), to recruit factors assisting in transactivation and to generate mRNAs within these CCLAs. The mRNAs are then exported into the cytosol for high-level protein expression. We have also displayed the surface antigens of the infectious viruses onto the envelope of baculovirus as a pseudovirus or onto the cytoplasmic membrane of the insect cells for functional analysis of these proteins. We found that such pseudoviruses can serve as a good vaccine for preventing virus infection and also could be a platform for sera detection when express these antigens on cells.
Our recent accomplishments are described as follows:
I. Baculovirus IE2 organize a clathrate cage-like apparatus as an active nanomachinery for gene transcription. (Wei et al., 2018. Manuscript submitted.)
Our previous study found that baculovirus IE2 can stimulate high level gene expression in mammalian cells. Since this is a very useful technology to boost gene activation for better baculovirus applications, therefore, we further study the mechanism behind such high level gene stimulation in exogenous hosts. We found that IE2 targets viral DNA indirectly through the Daxx and H3.3 proteins of host cells. IE2 then grows as a unique nuclear body, named clathrate cage-like apparatus (CCLA), and further recruits actin and RNA polymerase II to generate mRNAs within CCLAs. The actin and mRNAs are then exported into the cytosol for high-level protein expression.
This novel gene transactivation mechanism manipulated by IE2 is different from that of conventional gene activation, and it is significant for a number of reasons:
1. Transcription operates within a closed nuclear apparatus: The entire transactivation process is uniquely performed within CCLAs, with IE2 as the outer wall. IE2 CCLAs selectively recruit factors necessary for transactivation and simultaneously block those factors that inhibit it.
2. IE2 CCLAs is a highly functional nanomachinery: Spherical IE2 CCLAs recruit factors, including actin (Fig. 4) and RNA polymerase II, to generate mRNA, and then the mRNA is exported together with actin to the cytosol, resulting in hollow corpuscular CCLAs.

Fig. 4: IE2 forms CCLA (white arrow) nano-structure in the cell nucleus, which can recruit and export actin (green) for gene transactivation. Please see unique pictures that the loss of actin results CCLA from full (40 hours post transduction, hpt) into hollow (41.8 hpt) CCLA structures in the cell nucleus. It is very unique to see the in and out movements of actin, and later we showed that this activity is used to ship RNA out of CCLA.
3. IE2 CCLA is identified as an ancient yet conserved cellular transcriptional machinery in both insects and mammals: Since insects have diverged for more than 900 million years from mammals, the IE2 CCLA nanomachinery represents a very ancient cellular mechanism that is manipulated by viral IE2 for gene transactivation and yet remains functional in vertebrate systems.
4. This is a novel visible transcriptional system: Gene transactivation is conventionally deemed to be an invisible event. However, most if not all of the IE2 CCLA transactivation events were visible under light microscopy. Thus, it will provide a novel and convenient system for further studies of gene transactivation.
5. Our study represents one of the most complete investigations of nuclear foci: Proteins that form particles or foci in nuclei have been frequently reported in numerous publications. We believe our work represents one of the most complete studies on how a visible nuclear focus or body operates, although the mechanism by which IE2 works may be unique and different from other nuclear bodies.
This type of transactivation machinery is probably haven’t been found and should be an exciting and significant discovery.
II. Global screening of antiviral genes that suppress baculovirus transgene expression in mammalian cells. (Chang et al., Molecular Therapy - Methods & Clinical Development, 6: 194-206)
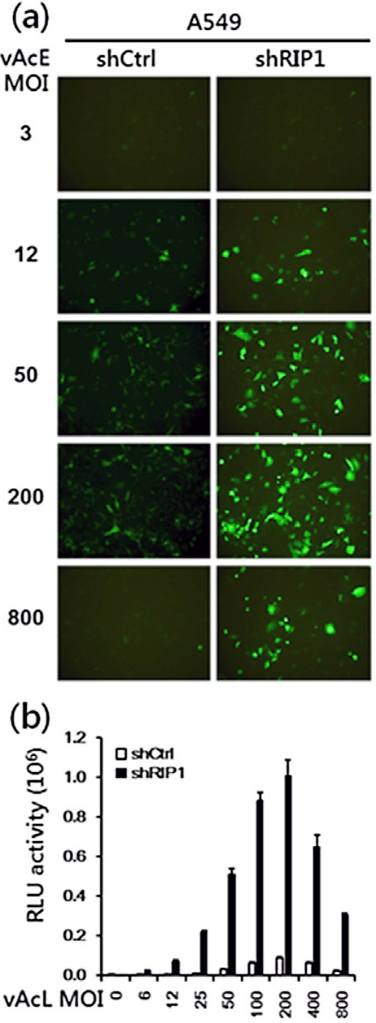
Although baculovirus has been used as a safe and convenient gene delivery vector in mammalian cells, baculovirus-mediated transgene expression is much less effective in these cell lines than in insect cells. Identification of host genes, which suppress baculovirus gene expression, is necessary to improve baculovirus-mediated expression systems. Here, we performed high throughput shRNA library screening, targeting 176 antiviral innate immune genes, and identified 43 host restriction factor genes in a human lung carcinoma cell line. Among them, suppression of receptor interaction protein kinase 1 (RIP1, alias RIPK1, Fig. 2) significantly increased baculoviral transgene expression without resulting in significant cell death. Silencing of RIP1 did not affect viral entry or cell viability, but it did inhibit nuclear translocation of IRF3 and NF-κB transcription factors. Also, activation of downstream signaling mediators (such as TBK1 and IRF7) was affected, and subsequent interferon and cytokine gene expression levels were abolished (Fig. 5). Further, Necrostatin-1 (Nec-1)—an inhibitor of RIP1 kinase activity—dramatically increased baculoviral transgene expression in RIP1-silenced cells. This is the first study of most of the known human cell antiviral innate immune response factors against a non-adaptive baculovirus. In addition, this study has made baculovirus a more efficient gene transfer vector for its better applications in the mammalian cell systems.
Figure 5. RIP1-knockdown (shRIP1) strongly elevates baculovirus transgene expression in non-host mammalian cells. Transgene (reporter gene) expression levels of stably-expressing shRNA for RIP1 (shRIP1) or control siRNA (shCtrl) in A549 cells transduced with vAcE (a) and vAcL (b) at various MOIs as indicated for 48 h. Reporter assay of EGFP expression (a) and luciferase expression (b) driven by a CMV promoter carried on the recombinant baculoviruses vAcE and vAcL.III. Identification of a high efficiency baculovirus DNA replication origin that functions in insect and mammalian cells. (Wu et al., 2014, Journal of Virology, 88:13073-85. Selected as a spot light paper by the journal)
We found that baculovirus, the Autographa californica multinucleocapsid nucleopolyhedrovirus (AcMNPV) p143 gene contains a novel replication origin (ori) in its coding region, which we later showed to be the strongest ori in the genome of this virus. This ori can enhance gene expression as driven by the polyhedrin promoter in insect cells and also the CMV promoter in the mammalian cells.
IV. Display of surface antigens from infectious viruses onto the envelope of baculovirus as effective vaccines. (Chang et al., Viruses, 2018, 10, 346-364; Chang et al., 2016, PLoS one, 11 (3): e0147485; Naik et al., 2018, Scientific Reports, under 2nd revision)
Human infectious viruses are all dangerous enveloped viruses. They are difficult to request from foreign countries, and special facilities, e.g. BSL3 laboratory, are needed to handle these viruses. These are all complex virus families, influenza viruses alone contains at least 18 different serotypes, and subtype viruses could be further divided within each serotypes. In year 2004, there was an urgent problem regarding what is the factor, which triggers cytokine storm and results human death by the SARS virus? We were that time displayed spike surface antigen from the SARS virus onto the envelope of baculovirus to maintain its trimeric structure, and used this baculovirus-based pseudovirus as a tool to identify and prove that the spike of SARS triggers cytokine storm (Chang et al., 2004). We have also displayed the spike protein of the porcine epidemic diarrhea virus (PEDV, a coronavirus) onto baculovirus as a carrier vaccine and prevent virus infection successfully. We also engineered hemagglutinin from the influenza virus on baculovirus as a vaccine to prevent the infection of influenza virus successfully. All these experiments proved that the carrier vaccine, where different foreign surface antigens are individually displayed on baculovirus, could be effective vaccines.
Copyright © 2017 IMBCC. All rights reserved. |
