大腦的發育仰賴一連串時序精準的轉換,而我們實驗室目前研究的是這些轉換在細胞與分子層次上如何被調控。我們研究神經元如何成熟、腦部關鍵期的可塑性如何開啟與關閉,以及中樞神經系統如何在面對刺激時仍維持適當的免疫耐受。我們的研究主要著重於三個彼此直接或間接連結的主題:蛋白質周轉的空間調控、發育中神經元的受力訊號傳遞,以及依賴神經活動的 RNA 剪接調控。透過先進顯微技術、模擬組織環境的培養系統,以及 CNS 免疫胜肽體學,我們希望了解這些機制如何共同塑造神經迴路發育與神經免疫平衡,並進一步連結正常生理與疾病狀態。
成熟軸突中的蛋白體運輸: 從長距離軸突運輸到神經膠細胞調控的免疫耐受
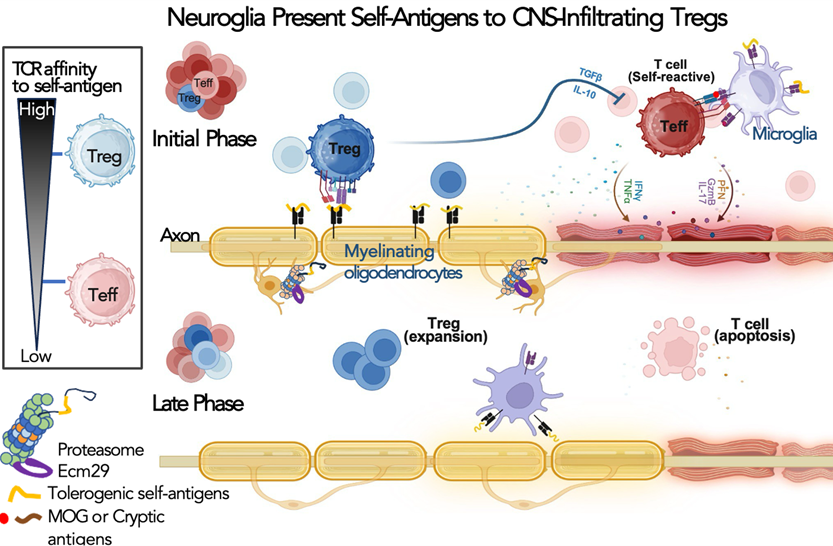
本實驗室研究蛋白酶體的空間調控,如何影響中樞神經系統中的神經發育與免疫耐受。我們的研究指出,蛋白酶體的運輸與定位會影響軸突發育、抑制性成熟的時序,以及神經興奮性。近年來,我們進一步發現,神經膠細胞中的 Ecm29/蛋白酶體依賴性抗原生成,會形塑 CNS 的自體抗原譜,並促進調節型 T 細胞反應。目前的研究聚焦於:特定自體抗原及其對應的 T 細胞受體,如何導向免疫耐受而非發炎,並希望建立神經發炎疾病中抗原特異性免疫調控的分子原理。
Illustration of our working model in which oligodendrocyte-presented tolerogenic self-antigens promote activation and expansion of CNS-infiltrating regulatory T cells.
從基質力學感知到關鍵期可塑性
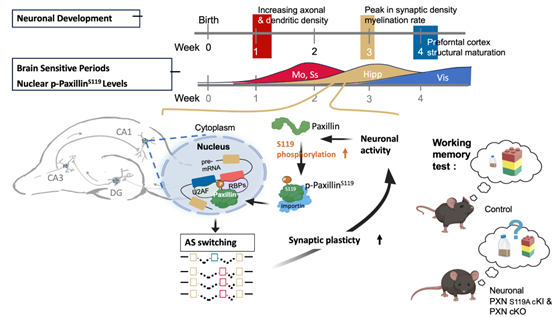
本實驗室的另一項核心研究主題,是研究發育中的神經元如何將機械訊號與神經活動訊號轉換為穩定的發育變化。透過模擬腦組織環境的培養系統,我們發現 paxillin 在神經突形成、受力依賴的導引,以及膜運輸調控中具有非典型功能。近年來,我們進一步鑑定 paxillin 為一種依賴神經活動的細胞核剪接調控因子,參與出生後關鍵期的腦部發育。目前的研究聚焦於不同 paxillin 磷酸化狀態如何協調細胞骨架與細胞核反應,從神經前驅細胞到成熟神經元,進而驅動腦部發育狀態的轉換。
Illustration of our working model in which neuronal activity induces paxillin phosphorylation and nuclear translocation during a critical developmental window, which in turn modulates alternative splicing programs required for synaptic function and short-term memory formation.
- PDF, 2006-2011, Dept. Mol. & Cell. Biol., UC Berkeley, USA
- Ph.D., 2004, Insti. Biochem., Natl. Yang Ming Univ.
- BS, 1998, Med. College, Med. Tech., Natl. Cheng Kung Univ.
- 2021, 國立成功大學優秀青年校友
- 2020, 科技部優秀青年學者研究計劃
- 2019, 中央研究院青年學者優秀著作獎
- 2019, Young Investigator Award for Pediatric Neuropsychiatric Science
- 2017–2022, Career Development Award, Academia Sinica
- 2004–2006, Studying Abroad Scholarship (教育部公費留學)
- Chu,† C.-H., G.-Z. Pan,† et al. (2025). Nuclear paxillin functions as a molecular switch for alternative splicing in neurons during a critical period of brain development. The EMBO Journal 44, 5965–5992. DOI: 10.1038/s44318-025-00560-8.
- Chen, S.-W., et al. (2025). ECM29/Proteasome-Mediated Self-Antigen Generation by CNS-Resident Neuroglia Promotes Regulatory T Cell Activation. Cell Reports 44, 115161. DOI: 10.1016/j.celrep.2024.115161.
- Chen, C., et al. (2022). Neuronal paxillin and drebrin mediate BDNF-induced force transduction and growth cone turning in a soft tissue-like environment. Cell Reports 40, 111188. DOI: 10.1016/j.celrep.2022.111188.
- Lee, M.,et al. (2020). Ecm29-mediated Proteasomal Distribution Modulates Excitatory GABA Responses in the Developing Brain. Journal of Cell Biology 219(2): e201903033. DOI: 10.1083/jcb.201903033.
- Chang, T.-Y., et al. (2017). Paxillin facilitates timely neurite initiation on soft-substrate environments by interacting with the endocytic machinery. eLife 6:e31101. DOI: 10.7554/eLife.31101.
- Hsu, M.-T., et al. (2015). Stage-dependent axon transport of proteasomes contributes to axon development. Developmental Cell 35, 418–431. DOI: 10.1016/j.devcel.2015.10.018.
This study identified paxillin as an activity-dependent nuclear regulator of alternative splicing during the postnatal critical period. It established a mechanistic link between synaptic activity, paxillin S119 phosphorylation, and critical-period splicing programs required for neural plasticity.
This work showed that ECM29/proteasome-dependent antigen generation in CNS-resident neuroglia helps shape the self-antigen landscape and promotes regulatory T cell activation. It extended my proteostasis research into CNS immune tolerance and neuroimmune regulation.
This study demonstrated that paxillin and drebrin form a softness-dependent module that mediates BDNF-induced force transduction and growth cone turning in soft brain-like environments. I led the project concept and integrated the mechanobiology, imaging, and developmental analyses.
This work showed that ECM29-dependent proteasome localization regulates local NKCC1 abundance, the timing of the excitatory-to-inhibitory GABA switch, and neuronal excitability during development. It extended my proteasome program from axon biology to functional circuit maturation.
This study identified a neuronal paxillin mechanism adapted to soft, brain-like mechanical environments and showed that paxillin promotes neurite initiation through interaction with the endocytic machinery. It established neuronal mechanobiology as a major direction of my independent research program.
This study identified ECM29 as a key adaptor linking proteasomes to long-range transport in developing neurons and showed that stage-dependent proteasome positioning contributes directly to axon development. It established the proteasome-distribution program as a major axis of my laboratory.