The brain develops through a series of precisely timed transitions, and our laboratory is interested in how those transitions are controlled at the cellular and molecular level. We study how neurons mature, how critical periods of plasticity open and close, and how the central nervous system maintains immune tolerance while remaining responsive to challenge. Our research focuses on three connected themes: spatial control of protein turnover, force-sensitive signaling in developing neurons, and activity-dependent regulation of RNA splicing. Using advanced imaging, tissue-mimetic culture systems, and CNS immunopeptidomics, we aim to understand how these processes work together to shape neural circuit development and neuroimmune balance in health and disease.
A. Proteasome-Controlled Homeostasis in the Central Nervous System: From Long-Range Axonal Transport to Neuroglia-Regulated Immune Tolerance
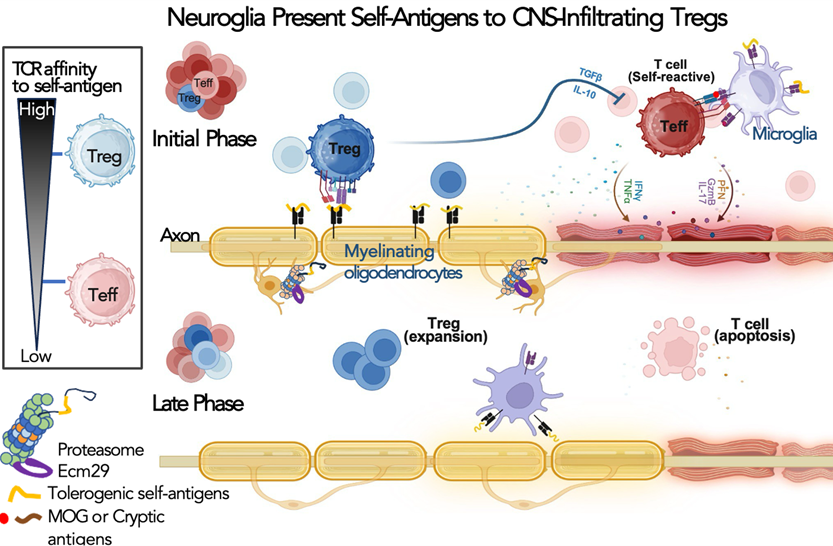
Our laboratory studies how spatial control of proteasomes regulates neuronal development and immune tolerance in the central nervous system. We showed that proteasome transport and localization influence axon development, the timing of inhibitory maturation, and neuronal excitability. More recently, we found that Ecm29/proteasome-dependent antigen generation in neuroglia helps shape the CNS self-antigen repertoire and promotes regulatory T cell responses. Our current work asks how selected self-antigens and their cognate T cell receptors drive immune tolerance rather than inflammation, with the goal of defining molecular principles for antigen-specific regulation in neuroinflammatory disease.
Illustration of our working model in which oligodendrocyte-presented tolerogenic self-antigens promote activation and expansion of CNS-infiltrating regulatory T cells.
B. Paxillin Moonlighting in Brain Development: From Substrate Mechanics Sensing to Critical-Period Plasticity
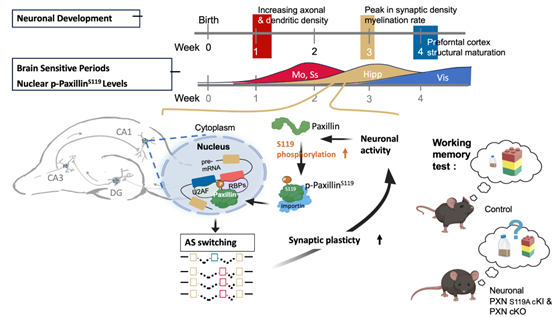
We investigate how developing neurons convert mechanical and activity-dependent signals into stable developmental changes. Using brain-mimetic culture systems, we discovered noncanonical roles of paxillin in neurite formation, force-dependent guidance, and membrane trafficking in soft tissue-like environments. More recently, we identified paxillin as an activity-dependent nuclear regulator of alternative splicing during the postnatal critical period. Our current work examines how distinct paxillin phosphorylation states coordinate cytoskeletal and nuclear responses across development, from neural progenitors to maturing neurons. This program aims to define paxillin as a mechanochemical regulator of developmental transitions in the brain.
Illustration of our working model in which neuronal activity induces paxillin phosphorylation and nuclear translocation during a critical developmental window, which in turn modulates alternative splicing programs required for synaptic function and short-term memory formation.
- PDF, 2006-2011, Dept. Mol. & Cell. Biol., UC Berkeley, USA
- Ph.D., 2004, Insti. Biochem., Natl. Yang Ming Univ.
- BS, 1998, Med. College, Med. Tech., Natl. Cheng Kung Univ.
- 2021, NCKU Distinguished Alumni Award
- 2020, Project for Excellent Junior Research Investigators, Ministry of Science and Technology
- 2019, Junior Research Investigator Award, Academia Sinica
- 2019, Young Investigator Award for Pediatric Neuropsychiatric Science
- 2017–2022, Career Development Award, Academia Sinica
- 2004–2006, Studying Abroad Scholarship (Ministry of Education)
- Chu,† C.-H., G.-Z. Pan,† et al. (2025). Nuclear paxillin functions as a molecular switch for alternative splicing in neurons during a critical period of brain development. The EMBO Journal 44, 5965–5992. DOI: 10.1038/s44318-025-00560-8.
- Chen, S.-W., et al. (2025). ECM29/Proteasome-Mediated Self-Antigen Generation by CNS-Resident Neuroglia Promotes Regulatory T Cell Activation. Cell Reports 44, 115161. DOI: 10.1016/j.celrep.2024.115161.
- Chen, C., et al. (2022). Neuronal paxillin and drebrin mediate BDNF-induced force transduction and growth cone turning in a soft tissue-like environment. Cell Reports 40, 111188. DOI: 10.1016/j.celrep.2022.111188.
- Lee, M.,et al. (2020). Ecm29-mediated Proteasomal Distribution Modulates Excitatory GABA Responses in the Developing Brain. Journal of Cell Biology 219(2): e201903033. DOI: 10.1083/jcb.201903033.
- Chang, T.-Y., et al. (2017). Paxillin facilitates timely neurite initiation on soft-substrate environments by interacting with the endocytic machinery. eLife 6:e31101. DOI: 10.7554/eLife.31101.
- Hsu, M.-T., et al. (2015). Stage-dependent axon transport of proteasomes contributes to axon development. Developmental Cell 35, 418–431. DOI: 10.1016/j.devcel.2015.10.018.
This study identified paxillin as an activity-dependent nuclear regulator of alternative splicing during the postnatal critical period. It established a mechanistic link between synaptic activity, paxillin S119 phosphorylation, and critical-period splicing programs required for neural plasticity.
This work showed that ECM29/proteasome-dependent antigen generation in CNS-resident neuroglia helps shape the self-antigen landscape and promotes regulatory T cell activation. It extended my proteostasis research into CNS immune tolerance and neuroimmune regulation.
This study demonstrated that paxillin and drebrin form a softness-dependent module that mediates BDNF-induced force transduction and growth cone turning in soft brain-like environments. I led the project concept and integrated the mechanobiology, imaging, and developmental analyses.
This work showed that ECM29-dependent proteasome localization regulates local NKCC1 abundance, the timing of the excitatory-to-inhibitory GABA switch, and neuronal excitability during development. It extended my proteasome program from axon biology to functional circuit maturation.
This study identified a neuronal paxillin mechanism adapted to soft, brain-like mechanical environments and showed that paxillin promotes neurite initiation through interaction with the endocytic machinery. It established neuronal mechanobiology as a major direction of my independent research program.
This study identified ECM29 as a key adaptor linking proteasomes to long-range transport in developing neurons and showed that stage-dependent proteasome positioning contributes directly to axon development. It established the proteasome-distribution program as a major axis of my laboratory.