Dendrite Development and Degeneration
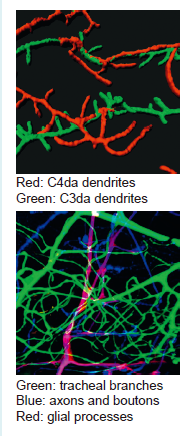
Neurons are the fundamental unit that makes up our brain. They are born of different types with distinct morphology, which is mostly accounted for by the dendritic trees. One fascinating question to us is how neurons grow such exuberant dendrites once they are born. We have been using Drosophila dendritic arborization (da) neurons as the model system to study dendrite development and degeneration. Among the four types of da neurons (classes I-IV), class IV da (C4da) neurons is most complex in terms of dendritic morphology. We envision that cellular machineries such as endocytosis and exocytosis are highly active during dendrite elaboration. Mainly by fluorescence imaging techniques and fly genetics, we investigate how various cellular processes such as actin dynamics, Golgi outpost transport, protein glycosylation and cell-cell interaction contribute to dendrite growth and degeneration.
We have explored a tripartite system to understand the interaction among synapses, glia and tracheal branches during development and physiology. The neuromuscular junctions that motor neurons form synapses with muscle cells are in particular of interest. The incoming axons are wrapped with glial processes till the terminals where they form bouton-like structures embedded in muscle membranes. Tracheal branches also innervate this area with higher density, presumably due to higher oxygen demands at NMJs. We have studied the effect of insufficient oxygen supply on the development of synapses, mediated by Wnt/Wg signal secreted from glial cells. In addition, glial cells secreted the micro-RNA miR-274 to coordinate the growth of tracheal branches and synaptic boutons during development. The full complements of tracheal branches and synaptic boutons likely contribute significantly to the hypoxia response behavior.
- PDF, 1993-1996, UC-San Francisco, USA
- Ph.D., 1993, Dept. Biochem. & Cell Biol., SUNY-Stony Brook, USA
- BS, 1985, Dept. Chemistry, Natl. Taiwan Univ.
- Postdoc. Fellowship, The Jane Coffin Childs Memorial Fund for Medical Research, 1994- 1997
- Academia Sinica Outstanding Young Scientist Award, 2000
- National Science Council Excellence in Research Award, 2001-2002, 2003-2005, 2009-2012
- National Science Council Frontier Research Grant, 2002-2007, 2007-2012, 2012-2017
- Academia Sinica Investigator Award, 2005-2009, 2010-2014
- Taiwan-France Science and Technology Award, 2009
- Ho Chin-Tui Foundation Award on Outstanding Contributions in Biology, 2013
- Yang, W.-K., Peng, Y.-H., Li, H., Lin, H.-C., Lin, Y.-J., Lai, T.-T., Suo, H., Wang, C.-H. Lin, W.-H., Ou, C.-Y., Zhou, X., Pi, H., Chang, H. C., Chien, C.-T. (2011) Nak regulates localization of clathrin sites in higher-order dendrites to promote local dendrite growth. Neuron. 72(2): 285-299.
- Lin, C.-H., Li, H., Lee, Y.-N., Cheng, Y.-J., Wu, R.-M., Chien, C.-T. (2015) Lrrk regulates the dynamic profile of dendritic Golgi outposts through the golgin Lava lamp. J. Cell Biol. 210(3): 471-483.
- Nithianandam, V., Chien, C.-T. (2018) Actin blobs prefigure dendrite branching sites. J. Cell Biol. 217: 3731-3746.
- Yang, W.-K., Chueh, Y.-R., Cheng, Y.-J., Siegenthaler, D., Pielage, J., Chien, C.-T. (2019) Epidermis-derived L1CAM homolog Neuroglian mediates dendrite enclosure and blocks heteroneuronal dendrite bundling. Curr. Biol. 29: 1445-1459.
- Chen, P.-Y., Tsai, Y.-W., Cheng, Y.-J., Giangrande, A., Chien, C.-T. (2019) Glial response to hypoxia in mutants of NPAS1/3 homolog Trachealess through Wg signaling to modulate synaptic bouton organization. PLoS Genet. 15: e1007980.
- Tsai, Y.-W., Sung, H.-H., Li, J.-C., Yeh, C.-Y., Chen, P.- Y., Cheng, Y.-J., Chen, C.-H., Tsai, Y.-C., Chien, C.-T. (2019) Glia-derived exosomal miR-274 targets Sprouty in trachea and synaptic boutons to modulate growth and responses to hypoxia. Proc. Natl. Acad. Sci. USA 116(49): 24651-24661.

