Orphan nuclear receptors recruit TRIM28 to promote telomeric H3K9me3 for the ALT pathway
Dr. Chen, Liuh-Yow - April, 2026
Cancer cells can use the alternative lengthening of telomeres (ALT) pathway to maintain telomere length, bypass cellular senescence, and achieve replicative immortality. However, the mechanism underlying the establishment of the key heterochromatin mark H3K9me3 at telomeres during ALT activation remains incompletely understood. In this study, orphan nuclear receptors were shown to recruit the transcriptional corepressor TRIM28 to telomeres, where it promotes H3K9me3 deposition and facilitates ALT activation. Mechanistically, orphan nuclear receptors recruit TRIM28 to telomeric chromatin through protein–protein interactions, thereby facilitating heterochromatin remodeling and functionally linking chromatin reorganization to ALT activation. These findings highlight the essential role of TRIM28-mediated heterochromatin formation in replicative immortality. This work was published on March 31, 2026, in The EMBO Journal by Dr. Liuh-Yow Chen’s group. The first author is Tsai Chia-Tsen, a PhD student in the TIGP-MCB program at Academia Sinica. The research was supported by Academia Sinica Grand Challenge Program and the National Science and Technology Council.
癌細胞永生的秘密:孤兒核受體透過TRIM28 啟動端粒延長機制
癌細胞會利用替代性端粒延長機制(Alternative Lengthening of Telomeres, ALT)維持端粒長度、避免細胞老化,進而維持細胞的複製永生性。然而,ALT 活化過程中關鍵的端粒異染色質標記 H3K9me3 的形成機制仍不完全清楚。本研究發現,孤兒核受體(orphan nuclear receptors)可透過招募轉錄共抑制因子 TRIM28,在端粒建立 H3K9me3 異染色質標記,進而促進 ALT 活化。本研究進一步指出,孤兒核受體透過蛋白質交互作用將 TRIM28 招募至端粒染色質,促進其參與異染色質重塑,並建立染色質重塑與 ALT 活化之間的功能性連結,突顯 TRIM28 所介導的異染色質標記在永生化細胞中的核心功能。 本研究成果由本所陳律佑副研究員團隊於 2026 年 3 月 31 日發表於《The EMBO Journal》,論文第一作者為中央研究院國際研究生學程「分子與細胞生物學」(TIGP-MCB)博士生蔡佳岑,研究經費由中央研究院關鍵突破計畫與國家科學及技術委員會支持。
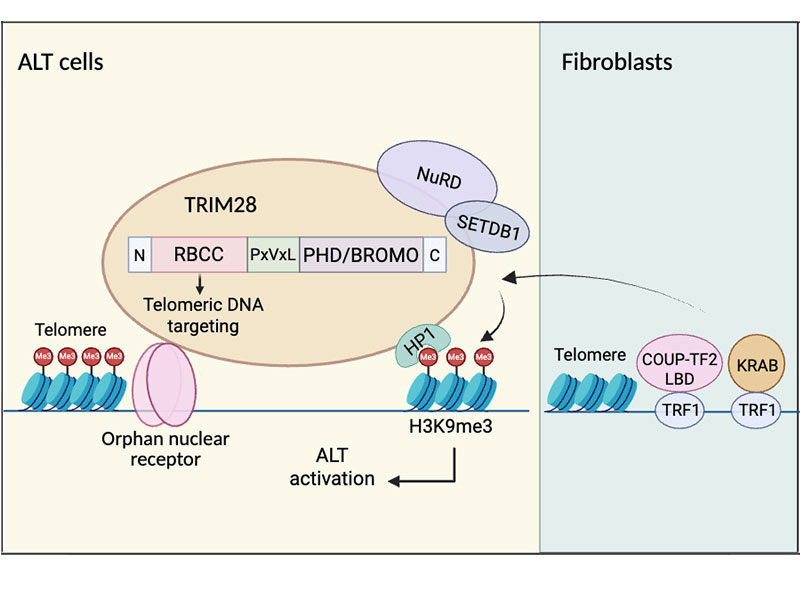
在癌細胞中,孤兒核受體與ALT端粒結合,並與TRIM28相互作用,從而維持H3K9me3並活化ALT細胞。在人類纖維母細胞中,孤兒核受體或KRAB轉錄抑制結構域與端粒的連接可透過TRIM28誘導H3K9me3和ALT表型。